《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 02 Structure and Properties of Organic Molecules

Organic Chemistry, ORGANIC 8th Edition CHEMISTRY L.G.Wade,Jr. Chapter 2 Lecture Structure and Properties of Organic Molecules .WAD E,JR Rizalia Klausmeyer Baylor University Waco,TX 2013 Pearson Education,Inc. ALWAYS LEARNING PEARSON
© 2013 Pearson Education, Inc. Chapter 2 Lecture Organic Chemistry, 8 th Edition L. G. Wade, Jr. Structure and Properties of Organic Molecules © 2013 Pearson Education, Inc. Rizalia Klausmeyer Baylor University Waco, TX

Wave Properties of Electrons nodal plane nucleus represented by nucleus wave function (instantaneous picture) nodal plane Standing wave vibrates in fixed location. Wave function,y,is a mathematical description of size,shape,and orientation. Amplitude may be positive or negative. Node:Amplitude is zero. 2013 Pearson Education,Inc. Chapter 2 2
© 2013 Pearson Education, Inc. Wave Properties of Electrons • Standing wave vibrates in fixed location. • Wave function, , is a mathematical description of size, shape, and orientation. • Amplitude may be positive or negative. • Node: Amplitude is zero. Chapter 2 2

Linear Combination of Atomic Orbitals Combining orbitals between two different atoms is bond formation. Combining orbitals on the same atom is hybridization. Conservation of orbitals Waves that are in phase add together. Amplitude increases. Waves that are out of phase cancel out 2013 Pearson Education,Inc. Chapter 2 3
© 2013 Pearson Education, Inc. Linear Combination of Atomic Orbitals • Combining orbitals between two different atoms is bond formation. • Combining orbitals on the same atom is hybridization. • Conservation of orbitals • Waves that are in phase add together. Amplitude increases. • Waves that are out of phase cancel out. Chapter 2 3

Sigma Bonding Electron density lies between the nuclei. A bond may be formed by s-s,p-p, s-p,or hybridized orbital overlaps. The bonding molecular orbital (MO)is lower in energy than the original atomic orbitals. The antibonding MO is higher in energy than the atomic orbitals. 2013 Pearson Education,Inc. Chapter 2 4
© 2013 Pearson Education, Inc. Sigma Bonding • Electron density lies between the nuclei. • A bond may be formed by s—s, p—p, s—p, or hybridized orbital overlaps. • The bonding molecular orbital (MO) is lower in energy than the original atomic orbitals. • The antibonding MO is higher in energy than the atomic orbitals. Chapter 2 4
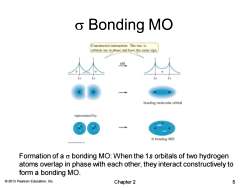
o Bonding MO Constructive interaction:The two 1s orbitals are in phase and have the same sign. add bonding molecular orbital represented by: a bonding MO Formation of a o bonding MO:When the 1s orbitals of two hydrogen atoms overlap in phase with each other,they interact constructively to form a bonding MO. 2013 Pearson Education,Inc. Chapter 2 5
© 2013 Pearson Education, Inc. s Bonding MO Formation of a s bonding MO: When the 1s orbitals of two hydrogen atoms overlap in phase with each other, they interact constructively to form a bonding MO. Chapter 2 5
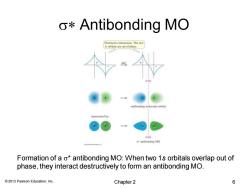
o*Antibonding MO Destructive interaction:The two 1s orbitals are out of phase. antibonding molecular orbital represented by: ot antibonding MO Formation of a o*antibonding MO:When two 1s orbitals overlap out of phase,they interact destructively to form an antibonding MO. 2013 Pearson Education,Inc. Chapter 2 6
© 2013 Pearson Education, Inc. s* Antibonding MO Formation of a s* antibonding MO: When two 1s orbitals overlap out of phase, they interact destructively to form an antibonding MO. Chapter 2 6
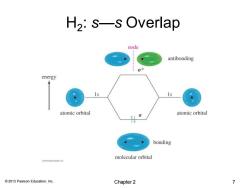
H2:s—s Overlap node antibonding 0端 energy atomic orbital atomic orbital 0 bonding molecular orbital 201 Pern Eaee 2013 Pearson Education,Inc. Chapter 2 7
© 2013 Pearson Education, Inc. H2 : s—s Overlap Chapter 2 7
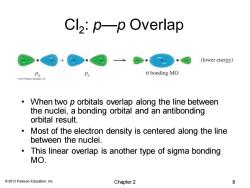
Cl2:p—p Overlap (lower energy) o bonding MO When two p orbitals overlap along the line between the nuclei,a bonding orbital and an antibonding orbital result. Most of the electron density is centered along the line between the nuclei. This linear overlap is another type of sigma bonding MO. 2013 Pearson Education,Inc. Chapter 2 8
© 2013 Pearson Education, Inc. Cl2 : p—p Overlap • When two p orbitals overlap along the line between the nuclei, a bonding orbital and an antibonding orbital result. • Most of the electron density is centered along the line between the nuclei. • This linear overlap is another type of sigma bonding MO. Chapter 2 8

Solved Problem 1 Draw the o*antibonding orbital that results from the destructive overlap of the two px orbitals just shown. Solution This orbital results from the destructive overlap of lobes of the two p orbitals with opposite phases.If the signs are reversed on one of the orbitals,adding the two orbitals gives an antibonding orbital with a node separating the two nuclei: node (higher energy) Px g来antibonding MO 0 Pearson Eacition ire 2013 Pearson Education,Inc. Chapter 2 9
© 2013 Pearson Education, Inc. Solved Problem 1 Draw the s* antibonding orbital that results from the destructive overlap of the two px orbitals just shown. This orbital results from the destructive overlap of lobes of the two p orbitals with opposite phases. If the signs are reversed on one of the orbitals, adding the two orbitals gives an antibonding orbital with a node separating the two nuclei: Solution Chapter 2 9
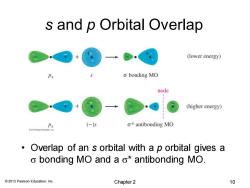
s and p Orbital Overlap (lower energy) o bonding MO node (higher energy) Px (-) o*antibonding MO 。 Overlap of an s orbital with a p orbital gives a o bonding MO and a o*antibonding MO. 2013 Pearson Education,Inc. Chapter 2 10
© 2013 Pearson Education, Inc. s and p Orbital Overlap • Overlap of an s orbital with a p orbital gives a s bonding MO and a s* antibonding MO. Chapter 2 10
按次数下载不扣除下载券;
注册用户24小时内重复下载只扣除一次;
顺序:VIP每日次数-->可用次数-->下载券;
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 01 Introduction and Review.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 26 Synthetic Polymers.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 25 Lipids.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 24 Amino Acids, Peptides, and Proteins.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 23 Carbohydrates and Nucleic Acids.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 22 Condensations and Alpha Substitutions of Carbonyl Compounds.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 21 Part 1 - Structure and Properties of Carboxylic Acid Derivatives.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 20 Carboxylic Acids.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 19 Amines.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 18 Ketones and Aldehydes.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 17 Reactions of Aromatic Compounds.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 16 Aromatic Compounds.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 14 Ethers, Epoxides, and Sulfides.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 13 Nuclear Magnetic Resonance Spectroscopy.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 12 Infrared Spectroscopy and Mass Spectrometry.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 11 Reactions of Alcohols.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 10 Structure and Synthesis of Alcohols.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 09 Alkynes.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 7th Edition, L. G. Wade, Jr.Pearson Education)Chapter 08 Reactions of Alkenes.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 03 Structure and Stereochemistry of Alkanes.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 05 Stereochemistry.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 06 Alkyl Halides - Nucleophilic Substitution and Elimination.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 07 Structure and Synthesis of Alkenes.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 08 Reactions of Alkenes.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 16 Aromatic Compounds.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Chapter 04 The Study of Chemical Reactions.ppt
- 《有机化学》课程PPT教学课件(Official PPT of Organic Chemistry, 8th Edition, L. G. Wade, Jr.Pearson Education)Reaction Mechanism and Synthesis Review.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 01 Structure and Bonding.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 02 Polar Covalent Bonds; Acids and Bases.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 03 Organic Compounds - Alkanes and Their Stereochemistry.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 04 Organic Compounds - Cycloalkanes and Their Stereochemistry.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 05 Stereochemistry at Tetrahedral Centers.pptx
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 06 An Overview of Organic Reactions.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 07 Alkenes and Alkynes.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 08 Reactions of Alkenes and Alkynes.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 09 Aromatic Compounds.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 11 Structure Determination - Nuclear Magnetic Resonance Spectroscopy.ppt
- 《有机化学》课程PPT教学课件(Organic Chemistry with Biological Applications, 3th Edition, John McMurry, 2016)Chapter 10 Structure Determination - Mass Spectrometry, Infrared Spectroscopy, and Ultraviolet Spectroscopy.ppt
